VISION
THT Biomaterials GmbH aims to overcome major methodological issues at the bottom of today’s drug development, tissue engineering and regenerative medicine research practices leading to false positive or negative results. A new visionary platform technology is under development that will speed up research as it makes its results more realistic by replacing animal components with human components.
Animal material free cell culture (3R)
- Higher impact for scientific publications in various research fields!
- Shortening the gap from research to patients in various clinical applications!!
Find all our products and services
The human placenta:
Animal material free cell culture (3R) from medical waste
Animal material free cell culture (3R) from medical waste

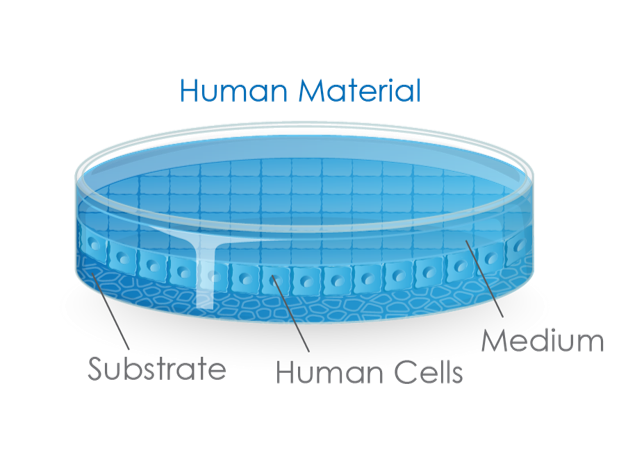
Petrischale Animal Material
Figure 1: the extensive use of animal materials in cell culture, starting point of most clinical developments, is still mostly performed with human cells, cultivated in animal materials, which do not represent our human model.
The Three Rs (3Rs) in relation to science are guiding principles for more ethical use of animals in testing. The 3Rs are:
Replace: substitute with alternative methods.
Reduce: decrease the number of laboratory animals.
Refinement: minimise the pain and suffering of laboratory animals.
Our approach to 3Rs is having a broader scope than simply encouraging alternatives to animal testing, we want to bring human material to human biology and medicine.
The human placenta:
Better quality because of the same species – we are not animals!
Better quality because of the same species – we are not animals!
Petrischale Human Material
Figure 2: the cultivation of human cells in combination with human proteins represent a closer model when compared to materials from other species. Human Placenta is medical waste, scalable, globally available, without any religious conflicts, no additional harm to donors, and a high support for donation for the parents and the society.
By using proteins from the same species you get:
Same (HUMAN) cell environment in vitro.
Less false positive/negative results.
Humanizing cell culture models.
More impact for researchers.
Faster into clinical applications
The human placenta:
A potential source for an affordable personalized medicine
A potential source for an affordable personalized medicine
Versatility and Compatibility
Figure 3: Human biomaterials may solve many research and clinical challenges over time, because till date, most cell culture models and medical products still comprise non-human components. Human placenta is biologically mostly derived from the human embryo, which would also enable around 500 g of autologous-raw-biomaterials for future-personalized-processing-by-patient/in case (our societies will change in the next decades).
Placenta is an organ largely composed of foetal elements (personalized medicine) and probably the most easily accessible raw tissue material without causing additional harm to donors. Many human placenta-based products have already been successfully used as topical or injectable agents in clinical approaches related to wound healing, burn injuries, post-surgical dressings or bedsores, for ophthalmology and other applications in regenerative medicine for over 100 years. We will use human-derived proteins and other biomaterials, to humanize cell culture in various research fields, comprising 2D and 3D applications such as material coatings, gels or tissue-inks. Moreover, human biomaterials are in favour when compared to biomaterials from other species in human patients such as (wound)filler, implants, haemostats, long-time-drug-deliveries, and many other clinical and aesthetical applications.
Our memberships
Austrian 3R Society (“Gesellschaft zur Förderung von Alternativen Biomodellen”)
EUSAAT (European Society for Alternatives to Animal Testing)
LISAvienna (life science austria)
ÖGMBT (Austrian Association of Molecular Life Sciences and Biotechnology)
ÖPPM (Österreichische Plattform für Personalisierte Therapie)
TERMIS (Tissue Engineering and Regenerative Medicine International Society)
The future of cell culture research and more
THT Biomaterials works closely with the pharmaceutical and health care industries as well as academic institutions and regulatory bodies to validate our HUMAN PLACENTA technology as approved and recognized method to replace animal materials in science. In close cooperation and constant dialogue with all stakeholders, our expert team designs and conducts the work effectively and with maximum transparency.
Our Business model
Placenta is donated with informed consent, transferred to THT Biomaterials, processed into human products, delivered to Universities, institutes and companies in a certified manner, used for high-impact scientific publications (=references for various applications), forcing THT Biomaterials to scale-up, searching for bigger pharma partners for further clinical developments into products, back to human patients (ideally, from the personalized “connector” towards the umbilical cord).
How we do it
We are driven by independent specialist with various backgrounds in different fields, an excellent network around, and we can act very fast, agile, and flexible.
